|
3/1/2024 0 Comments Water and potassium reaction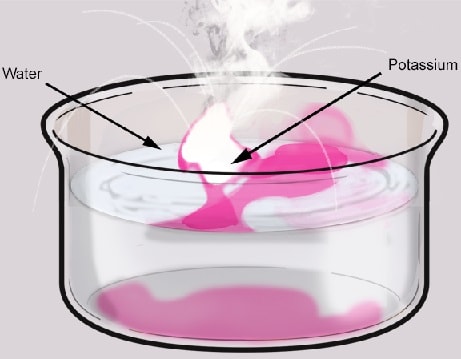 
They could write the equation in chemical. They could write a word equation for the chemical reaction: 'Two molecules of hydrogen gas react with one molecule of oxygen gas to produce two molecules of water vapor.' 3. They could draw a picture of the chemical reaction. To balance the chemical equation, begin by multiplying the element or compound which has less number of atoms with a number that would make the number equal on both sides. Chemists have a choice of methods for describing a chemical reaction. Balancing of the reaction by hit and trial method:Ī chemical equation is said to be balanced when atoms of every element in the chemical equation are equal in number on both (reactant and product) sides. K 2 O + H 2 O → KOH ( Potassium oxide ) ( Water ) ( Potassium hydroxide )Ģ. Complete the reaction by writing the products: The reaction of Potassium oxide with water gives Potassium hydroxide and the complete chemical equation is: K(s) + O 2 (g) KO 2 (s) Reaction with Water: Potassium reacts rapidly and vigorously with water to form potassium hydroxide. When potassium is burned in air, potassium superoxide (KO2) is formed. Complete the reaction by writing the products.ġ. Reaction with air: Though potassium is shiny when it’s cut, the reaction with air and moisture present in it tarnishes it.The alkali metals also include lithium, sodium, rubidium, cesium, and francium. The periodic table is a chart that shows how chemical elements are related to one another. JEE Main 2022 Question Paper Live Discussion The alkali metals are the elements that make up Group 1 (IA) of the periodic table.The heat produced by this reaction may ignite the. The balanced chemical reaction can be written as: 2 K ( s) Potassium + 2 H 2 O ( l) Water 2 KOH ( l) Potassium hydroxide + H 2 ( g) Hydrogen + Heat. Alkali metals react with water to produce heat, hydrogen gas, and the corresponding metal hydroxide. So, this is an example of an Exothermic reaction. 
Difference Between Selling And Marketing When Potassium reacted with water it produces potassium hydroxide and hydrogen with a large amount of heat.TS Grewal Solutions Class 11 Accountancy Potassium reacts violently with water to produce half a mole of hydrogen per mole of potassium and water and generates approximately 47 kilocalories per mole of heat.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.NCERT Solutions For Class 6 Social Science.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 8 Social Science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
